Water is added to produce a slurry or pulp. 
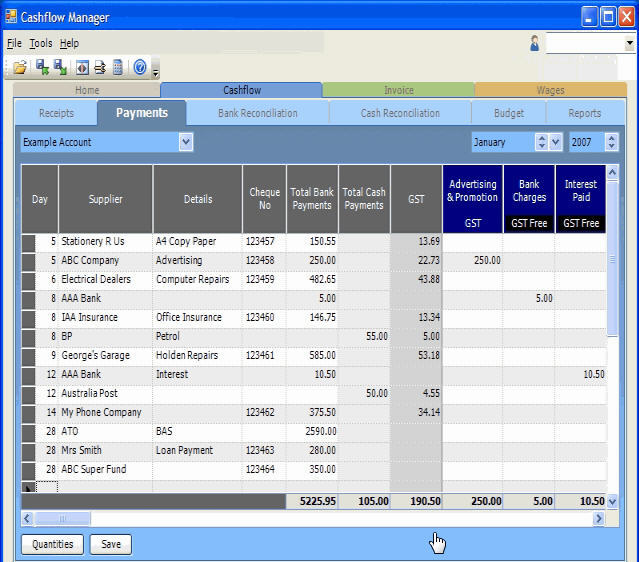
Depending on the ore, it is sometimes further concentrated by froth flotation or by centrifugal (gravity) concentration. The ore is comminuted using grinding machinery. from which it can be recovered by adsorption onto activated carbon. The soluble gold species is dicyanoaurate. Gold is one of the few metals that dissolves in the presence of cyanide ions and oxygen. Potassium cyanide and calcium cyanide are sometimes used in place of sodium cyanide. The chemical reaction for the dissolution of gold, the "Elsner equation", follows:Ĥ Au + 8 NaCN + O 2 + 2 H 2O → 4 Na + 4 NaOH Chemical reactions Ball-and-stick model of the aurocyanide or dicyanoaurate(I) complex anion, − Cyanide leaching "heap" at a gold mining operation near Elko, Nevada Their process is the Merrill–Crowe process. Īround 1900, the American metallurgist Charles Washington Merrill (1869–1956) and his engineer Thomas Bennett Crowe improved the treatment of the cyanide leachate, by using vacuum and zinc dust. Peyton had refined the process at his Mercur Mine in Utah, "the first mining plant in the United States to make a commercial success of the cyanide process on gold ores." In 1896, Bodländer confirmed that oxygen was necessary for the process, something that had been doubted by MacArthur, and discovered that hydrogen peroxide was formed as an intermediate. The process was first used on the Rand in 1890 and, despite operational imperfections, led to a boom of investment as larger gold mines were opened up. 
By suspending the crushed ore in a cyanide solution, a separation of up to 96 percent pure gold was achieved. Several patents were issued in the same year. In 1887, John Stewart MacArthur, working in collaboration with brothers Robert and William Forrest for the Tennant Company in Glasgow, Scotland, developed the MacArthur–Forrest process for the extraction of gold from gold ores. The gold could not be extracted from this compound with any of the then available chemical processes or technologies. The expansion of gold mining in the Rand of South Africa began to slow down in the 1880s, as the new deposits being found tended to contain pyritic ore. Industrial process John Stewart MacArthur developed the cyanide process for gold extraction in 1887. the stoichiometry of the soluble compound. Through the work of Bagration (1844), Elsner (1846), and Faraday (1847), it was determined that each atom of gold required two cyanide ions, i.e. In 1783, Carl Wilhelm Scheele discovered that gold dissolved in aqueous solutions of cyanide. 
At industrial scale, pH control is mainly achieved using lime, as an important enabling reagent in gold processing. A key feature for safe use of cyanide is to ensure adequate pH control at an alkaline pH level above 10.5. Cyanide can be safely used in the gold mining industry. Due to the highly poisonous nature of cyanide, the process is controversial and its use is even banned in some parts of the world. Other metals are recovered from the process include copper, zinc, and silver, but gold is the main driver of this technology. Production of reagents for mineral processing to recover gold represents more than 70% of cyanide consumption globally. Cyanidation is also widely used in the extraction of silver, usually after froth flotation. It is the most commonly used leaching process for gold extraction. Gold cyanidation (also known as the cyanide process or the MacArthur–Forrest process) is a hydrometallurgical technique for extracting gold from low-grade ore by converting the gold to a water-soluble coordination complex. Technique for extracting gold from low-grade ore
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
